University of Maryland, College Park
Department of Cell Biology and Molecular Genetics
The Andrews Research Group
Current Research
We pursue two lines of investigation – the cell biology of the intracellular protozoan parasites, and mechanisms of plasma membrane repair. These two lines of work, although independent and carried out by different members of the lab, often intersect. In fact, our interest in the mechanism of plasma membrane repair, a fundamental cell biological process, was born from our studies of how the intracellular parasite Trypanosoma cruzi invades host cells. We found that T. cruzi increases the intracellular calcium concentration in host cells, a signaling process that triggers recruitment and fusion of lysosomes with the plasma membrane.
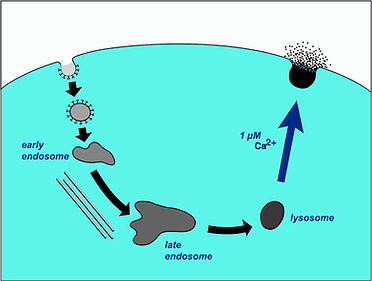
Our early studies with T. cruzi led to the discovery that conventional lysosomes can behave as calcium regulated secretory vesicles, which fuse with the plasma membrane releasing their contents when the cytosolic calcium concentration rises above 1 micromolar.
Plasma Membrane Repair
When wounded, eukaryotic cells reseal their plasma membrane (PM) within seconds. Earlier studies showed that PM repair involves exocytosis of intracellular vesicles, triggered by the influx of extracellular calcium into wounded cells. Our lab then showed that peripheral lysosomes correspond to the vesicles responsible for this fast exocytic response. In recent years we found that release of a key lysosomal enzyme, acid sphigomyelinase, triggers a massive and rapid form of endocytosis that participates in removing lesions from the PM. We are currently characterizing this novel form of endocytosis and investigating how lysosomal hydrolases secreted by wounded cells influence this process.
Check out Norma Andrews' iBiology talk here!
Intracellular Parasites
We are currently focusing our attention on Leishmania, an intracellular parasite of macrophages that is the agent of serious infections throughout the world. We have been investigating how these parasites subvert host cell function to get access to key nutrients, such as iron. In recent years we have identified several Leishmania membrane proteins that mediate iron and heme acquisition, and also showed how these parasites block iron export from macrophages for their own benefit. Surprisingly, in the course of these studies we discovered that iron uptake also regulates the signaling mechanism controlling the differentiation of Leishmania into amastigotes, infective forms capable of replicating in macrophages. We are characterizing several Leishmania proteins that are upregulated under conditions of iron and heme deprivation, and investigating how these parasite molecules interact/compete with equivalent iron and heme transporters in host cells.